Epigenetic mechanisms controlling cell fate decisions during the early stage of liver, pancreas and biliary tree development
Prof. Dr. Ludovic Vallier
The overarching goal of the Vallier group based at the Max Planck Institute for Molecular Genetics (MPIMG), is to understand liver organogenesis and to use the resulting knowledge for the development of new therapies. For that, we combine human induced Pluripotent Stem Cells (hiPSCs), primary organoids, gain and loss of function approaches and single-cell analyses to study human liver development in vitro. Of particular interest, we aim to uncover (i) the molecular mechanisms controlling the specification of hepatocytes and cholangiocytes, (ii) the factors driving the functional maturation of the same cell types and (iii) how these mechanisms relate to tissue regeneration. We then exploit this knowledge to generate cells for disease modelling and for cell-based therapy. This translational activity is currently located in the Vallier lab based at the Berlin Institute of Health Centre for Regenerative Therapy (BCRT).
We are looking for an experimental doctoral candidate to join our team at the MPIMG. Computational knowledge is always helpful, but bioinformatics training can also be provided. The successful candidate will study the epigenetic mechanisms controlling cell fate decisions during the early stage of liver, pancreas and biliary tree development. We are particularly interest to understand how these early steps control cellular diversity and plasticity in adult tissue in health and in disease.
For some of our recent work please refer to:
- Brevini et al., “FXR inhibition may protect from SARS-CoV-2 infection by reducing ACE2”. Nature. 2023. doi: 10.1038/s41586-022-05594-0. PMID: 36470304.
- Wesley et al., “Single-cell atlas of human liver development reveals pathways directing hepatic cell fates”. Nat Cell Biol.2022. 24(10):1487-1498. doi: 10.1038/s41556-022-00989-7. Epub 2022 Sep 15. PMID: 36109670.
- Sampaziotis et al., “Cholangiocyte organoids can repair bile ducts after transplantation in the human liver”. Science 2021. 371(6531):839-846. doi: 10.1126/science.aaz6964. PMID: 33602855.
For more information visit the Vallier’s lab websites:
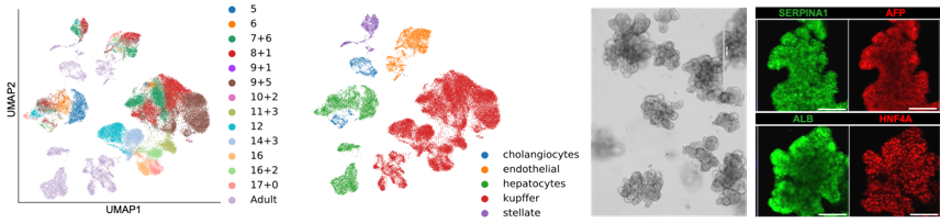
Single-cell map of human liver development and derivation of primary hepatoblast organoids (HBOs). Right: UMAP analyses of the developing human liver. Left: Bright field image of human HBOs and immunostaining showing expression of hepatoblast markers in HBOs. (Carola Morell; Wesley et al., Nature Cell Biology, 2022).



