Transcriptional condensates
Mammalian cells contain several hundred large clusters of transcriptional enhancers referred to as super-enhancers or stretch enhancers, which control genes that have especially prominent roles in cell-type-specific processes. Super-enhancers are occupied by an unusually high density of interacting factors, and are exceptionally vulnerable to perturbation of components commonly associated with most enhancers.
Compartmentalization of biochemical reactions often occurs in biomolecular condensates, which are membraneless bodies within the cytosol or nucleus. These condensates, e.g. nucleoli, Cajal bodies, nuclear speckles or DNA damage foci are formed as a result of liquid-liquid phase separation. Components are highly concentrated within condensates, and the formation and dissolution of condensates occurs with sharp transitions.
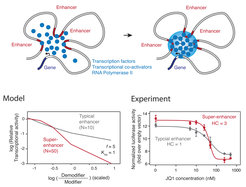
Based on the similar features of biomolecular condensates and super-enhancers, we recently proposed a model that a phase-separated multi-molecular assembly underlies the formation and function of super-enhancers. We are conducting experimental tests of this model, and explore the predictions of this model in diverse cellular contexts. We develop approaches to assess the functional importance of phase separation in gene control during differentiation and tumorigenesis.
Further reading:
Hnisz et al., Cell 2017
Whyte et al., Cell 2013
Hnisz et al., Cell 2013