4D Genome Architecture, Gene Regulation and Developmental Diseases
Most developmentally important genes have complex and pleiotropic expression patterns. Such spatially and temporally restricted transcriptional activities are controlled by cis-regulatory elements (enhancers, promoters) that can be far away from their target gene. To date, little is known about how enhancers contact and activate and their target genes. Specifically, how the genome functions within the space of the nucleus and its dynamic context have remained elusive and are probably one of the biggest challenges in the post-genomic era.
In the last few years, the newly developed Hi-C methods have shown that the genome is partitioned in megabase scale compartments called topologically associated domains (TADs). Because TADs restrict the range of enhancer targets, enhancers usually contact genes located within these TADs, but not outside. TADs have been shown to be surprisingly stable across cells, tissues and even species, suggesting that they function as a general folding scaffold determining domains of possible interaction partners.
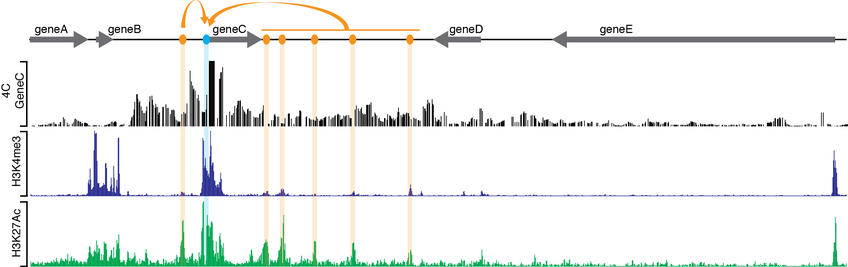
Using limb development as a model system, we are investigating changes in 3D genome organisation during development. To do so, we use a large variety of state-of-the-art technologies: on the one hand, we employ Chromosomal Conformation Capture technologies (4C, Hi-C), ChIP-seq, and RNA-seq to describe regulatory landscapes. On the other hand, we apply CRISPR-Cas9 genome editing to induce various genetic re-arrangements (deletions, inversions, duplications) and study their impact on chromatin architecture, gene regulation, and limb development.
1.
Kragesteen BK, Spielmann M, Paliou C, Heinrich V, Schöpflin R, Esposito A, Annunziatella C, Bianco S, Chiariello AM, Jerković I, Harabula I, Guckelberger P, Pechstein M, Wittler L, Chan WL, Franke M, Lupiáñez DG, Kraft K, Timmermann B, Vingron M, Visel A, Nicodemi M, Mundlos S, Andrey G.
Dynamic 3D chromatin architecture contributes to enhancer specificity and limb morphogenesis.
Nat Genet. 2018 Oct;50(10):1463-1473. Epub 2018 Sep 27.
2.
Spielmann M, Lupiáñez DG, Mundlos S.
Structural variation in the 3D genome.
Nat Rev Genet. 2018 Jul;19(7):453-467. Review.
3.
Bianco S, Lupiáñez DG, Chiariello AM, Annunziatella C, Kraft K, Schöpflin R, Wittler L, Andrey G, Vingron M, Pombo A, Mundlos S, Nicodemi M.
Polymer Physics Predicts the Effects of Structural Variants on Chromatin Architecture
Nat Genet. 2018 May;50(5):662-667. Epub 2018 Apr 16.
4.
Andrey G, Mundlos S.
The three-dimensional genome: regulating gene expression during pluripotency and development.
Development. 2017 Oct 15;144(20):3646-3658. Review.
5.
Will AJ, Cova G, Osterwalder M, Chan W-L, Wittler L, Brieske N, Heinrich V, de Villartay J-P, Vingron M, Klopocki E, Visel A, Lupiáñez DG, Mundlos S.
Composition and dosage of a multipartite enhancer cluster control developmental expression of Ihh (Indian hedgehog).
Nat Genet. 2017 Oct;49(10):1539-1545. Epub 2017 Aug 28.
6.
Franke M, Ibrahim DM, Andrey G, Schwarzer W, Heinrich V, Schöpflin R, Kraft K, Kempfer R, Jerković I, Chan WL, Spielmann M, Timmermann B, Wittler L, Kurth I, Cambiaso P, Zuffardi O, Houge G, Lambie L, Brancati F, Pombo A, Vingron M, Spitz F, Mundlos S.
Formation of new chromatin domains determines pathogenicity of genomic duplications.
Nature 2016 Oct 13;538(7624):265-269. Epub 2016 Oct 5.
7.
Lupiáñez DG, Spielmann M, Mundlos S.
Breaking TADs: How Alterations of Chromatin Domains Result in Disease.
Trends Genet. 2016 Apr;32(4):225-237. Epub 2016 Feb 7.
8.
Lupiáñez DG, Kraft K, Heinrich V, Krawitz P, Brancati F, Klopocki E, Horn D, Kayserili H, Opitz JM, Laxova R, Santos-Simarro F, Gilbert-Dussardier B, Wittler L, Borschiwer M, Haas SA, Osterwalder M, Franke M, Timmermann B, Hecht J, Spielmann M, Visel A, Mundlos S.
Disruptions of Topological Chromatin Domains Cause Pathogenic Rewiring of Gene-Enhancer Interactions.
Cell. 2015 May 21;161(5):1012-1025. Epub 2015 May 7.
9.
Kraft K, Geuer S, Will AJ, Chan WL, Paliou C, Borschiwer M, Harabula I, Wittler L, Franke M, Ibrahim DM, Kragesteen BK, Spielmann M, Mundlos S, Lupiáñez DG, Andrey G.
Deletions, Inversions, Duplications: Engineering of Structural Variants using CRISPR/Cas in Mice.
Cell Rep. 2015 Feb 4. pii: S2211-1247(15)00029-7. [Epub ahead of print]
10.
Ibn-Salem J, Köhler S, Love MI, Chung HR, Huang N, Hurles ME, Haendel M, Washington NL, Smedley D, Mungall CJ, Lewis SE, Ott CE, Bauer S, Schofield PN, Mundlos S, Spielmann M, Robinson PN.
Deletions of chromosomal regulatory boundaries are associated with congenital disease.
Genome Biol. 2014 Sep 4;15(9):423.