Decisions at the crossroads of cellular life
A barrier is established shortly after stem cells differentiate
Why is it possible to reprogram committed body cells to become stem cells again? In search of an answer to this question, Alexander Meissner and colleagues investigated the transition phase between the stem cell state and specialized cells that identified a molecular barrier that controls the efficiency of reprogramming. The results appear in Nature Communications.
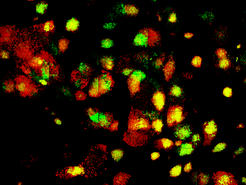
In the transition phase, most of the differentiating cells return to their stem cell state as soon as the OSKM factors have been reactivated. Under the microscope, stem cell colonies appear green, while all other cells remain red.
That is life: The fertilized egg initially develops into an embryo with indeterminate pluripotent cells, which are then able to generate the whole organism. “In nature, this process is a one-way street”, says Dr. Alexander Meissner, Director at the Max Planck Institute for Molecular Genetics in Berlin and Head of the Department of Genome Regulation.
The possibility to reverse the cell’s path from being an all-rounder that turns into highly specialized liver, muscle or skin cells, has been known for more than 50 years, explains Meissner. “It’s just that in 2006, the process suddenly became much easier.” At the time, the Japanese scientist Shinya Yamanaka succeeded in tricking mature skin cells directly into what he termed induced pluripotent stem cells (iPS cells). Meissner, who was working in this field at the time, was fascinated and focused his subsequent studies at Harvard University and now at the Max Planck Institute for Molecular Genetics in Berlin on understanding this remarkable process.
Just like embryonic stem cells, the iPS cells can easily turn into practically any type of cell. “iPS cells need only a few days to differentiate into a somatic cell type,” says Meissner. “We were curious to explore when that decision is final and the cells can no longer turn back.” In a new study recently published in the journal Nature Communications, the scientist and his team show when it becomes difficult to reverse this commitment and explore the underlying mechanism.
Reprogramming is still a black box for researchers
This work spanned many years of research and a move across the Atlantic, explains the scientist. He points to a small, red-framed picture on the wall of his office. It shows a floor plan of his first laboratory he headed in the department of Stem Cell and Regenerative Biology at Harvard University. His colleagues scribbled their signatures and farewell wishes all over it. It is where the study began with the question: What are the molecular changes stem cells undergo while specializing into body cells?
“The mechanisms underlying the reprogramming of cells are still largely a black box for us,” says Meissner. “From the perspective of developmental biology, it is unclear why all the cells of the body maintain the potential for reprogramming at all.” Because in normal development, the pluripotent stem cells “differentiate” into mature cells and these are not supposed to change their identity anymore. From a liver cell, only more liver cells arise, and the same goes for any cell like muscle cells or skin cells.
Observing changes before and after differentiation
Camille Sindhu and Zack Smith from Meissner's team at Harvard started working on the experiments together with Evgeny Yurkovsky from the Iftach Nachman lab at Tel Aviv University. They first generated iPS cells from connective tissue cells of a mouse by activating a group of genes famously called “OSKM”: Oct4, Sox2, Klf4 and c-Myc. These contain the blueprints for transcription factor proteins that are active only in stem cells and control the stem cell program. They trigger a cascade of molecular processes that can plunge already differentiated cells into an “identity crisis”.
After two weeks, the factors transform one out of a hundred body cells into a pluripotent stem cell. This cell keeps dividing, but does not differentiate as long as its nutrient solution is supplemented with hormone-like signal substances that act as “stem cell signals”.
Later the team was expanded by Sudhir Thakurela, a computational biologist in the Meissner lab at Harvard and Christina Riemenschneider, an experimental biologist in the lab in Berlin. They removed the stem cell signals in the culture medium and observed as the cells slowly turned into differentiated cells. They then kept checking whether and how well they were able to switch back to the stem cell state, as soon as the cells received just the stem cell signal or in combination with the four OSKM factors.
In a suspended state between stem and body cell
In the experiment, Meissner’s team slid back and forth between the stem cell and specialized body cell states, thereby focusing on the period in the life of the cell, in which it ponders a certain “career path”, but has not yet fully embarked on it. This transition phase is about 48 hours long, the scientists found. During this phase, the differentiating cells quickly and efficiently returned to their stem cell status, as soon as the researchers re-activated the OSKM factors for 12 to 24 hours. Up to 80 percent of cells became stem cells again.
The success rate fell to less than one percent as soon as the scientists gave the cells more than the two days to develop. By then, the cells had become already so highly specialized that reprogramming efforts took the usual two weeks, meaning whether the cells were differentiated for just days or came from mature body cells no longer played a role.
Back into the stem cell state
It only takes a few hours for differentiating cells to turn into pluripotent stem cells again under the influence of OSKM factors. Under the microscope, the stem cell cell colonies appear green, while all other cells remain red.
An identity-creating tipping point
As soon as the cells crossed this particular window, the stem cell program could no longer be efficiently re-activated. Meissner’s team was particularly interested in this tipping point and wanted to figure out, what changes from before and after and what exactly defines this transition phase.
The researchers profiled the behavior of the factor Oct4 during this critical phase. As a member of the OSKM group it is indispensable for the reprogramming. The powerful transcription factor controls a variety of genetic programs by selectively docking to different sites on the DNA strand activating thousands of genes.
The way back to indecision
In cells that can still efficiently return to the stem cell state, Oct4 accessed a subset of DNA regions that it usually binds in the stem cells. In total, there are about 3,000 accessible regions in the genome. These sites or at least a subset thereof might be key to pave the way for reactivating the entire stem cell program, the researchers concluded.
Interestingly, some of these DNA sites retain an “epigenetic memory” of the pluripotent state even in the fully differentiated body cells. Usually, cells block out unneeded developmental genes and marks them inactive after differentiation. “Here a small subset of genomic sites appears to remain open to Oct4 in all body cells,” says Meissner. The researcher can only guess, why the cell does not also epigenetically block these sections: “It might have something to do with the development of the gametes, which are specified at a later point in mammalian development.”
During fertilization, the gametes, i.e. ova and sperm, unite to form the first cell of a new organism. They have some regulatory properties that resemble stem cells. After all, they are supposed to develop into a completely new organism. “Maybe the cell cannot afford to epigenetically lock down the stem cell program completely, because the organism has not yet decided which cells should go into the germ line," says the scientist.
“Perhaps the germ line program enables cellular reprogramming – as a sort of side effect,” speculates Meissner. This, however, would have to be shown in further studies. Although the publication sheds some light on the “black box” of reprogramming, the exact interplay of genes during the cell’s most important career decisions remains opaque.
[blk]












